Wire Mesh for Hydrogen Production, Using Electrolysis
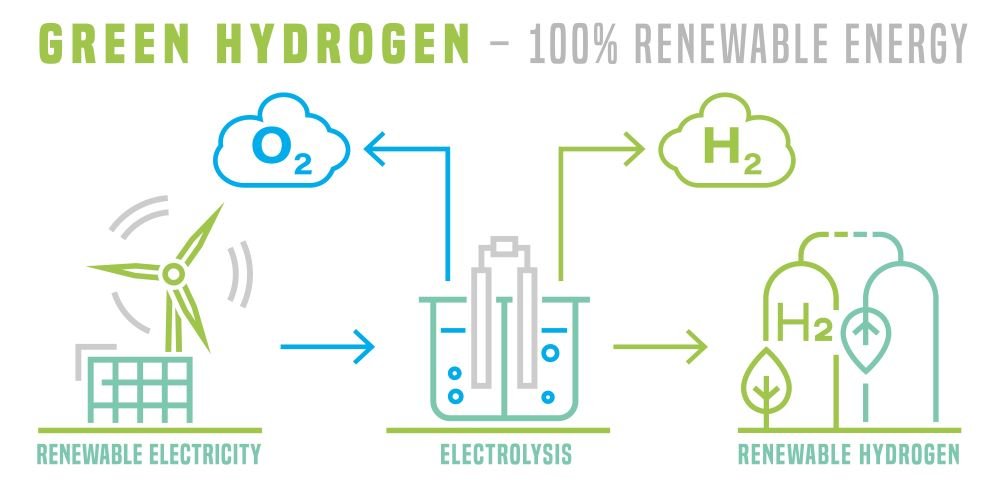
Wire mesh can be used in several ways in hydrogen production, but one of the most common ways is in the construction of electrodes for electrolysis cells. Electrolysis is the process of splitting water into hydrogen and oxygen using an electric current. In an electrolysis cell, two electrodes are placed in a solution of water and an electrolyte, and an electric current is passed through the solution. The electrodes are typically made of a conductive material, such as platinum or graphite, and are often coated with a layer of catalyst material to enhance the reaction.
Wire Mesh for Electrode Construction
Wire mesh can be used as the substrate for the electrode, onto which the catalyst material is coated. The wire mesh provides a high surface area for the catalyst material, which allows for a greater reaction rate and efficiency. Additionally, the wire mesh provides mechanical strength and support to the electrode, which is important in maintaining the integrity of the cell over time.
Wire Mesh for Gas Diffusion Layers
Another way that wire mesh can be used in hydrogen production is in the construction of gas diffusion layers (GDLs) in fuel cells. A GDL is a porous material that allows reactant gases to diffuse into the catalyst layer of the fuel cell while also allowing products and water to be removed. The wire mesh can be used as the backbone of the GDL, providing mechanical strength and support to the porous material. The wire mesh can also be coated with a hydrophobic material to prevent water from accumulating in the GDL, which can impede reactant gas diffusion and decrease cell performance.
Summary
Overall, wire mesh plays an important role in hydrogen production by providing mechanical strength and support to electrodes and GDLs, while also providing a high surface area for catalyst material to enhance reaction rates and efficiency.
